Opioid Prescription Labels Must Include Information About Naloxone: FDA
The US Food and Drug Administration (FDA) has issued a statement that all opioid drug makers must add recommendations about naloxone to their patients’ prescribing information. The new labeling change is being made to lower the risk of death due to fatal opioid overdose by making naloxone more available to those who need it.
What is Naloxone?
Opioids act on the body’s nervous system and cause it to slow down. In high enough doses, opioid use can cause the respiratory system to slow down or stop entirely.
Naloxone is a medication given to temporarily stop the effects of opioid drugs in the body. It can help restore respiration in someone who is experiencing an opioid overdose.
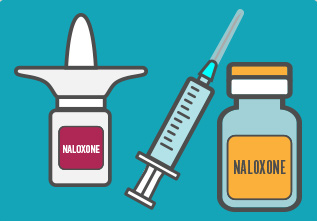
Naloxone can be administered using three methods: nasal spray, by injection, or by auto-injector. Its effects last for 20-30 minutes. Emergency services should always be called when naloxone has been administered. The patient needs to be carefully monitored afterward.
FDA Recommends Doctors Discuss Opioid Prescription Overdose Risk with Patients
The FDA is recommending that doctors writing prescriptions for opioid pain medications or medications to treat OUD (opioid use disorder) should discuss naloxone and where to obtain it with their patients. It is also recommending that doctors consider prescribing naloxone to patients who are at risk of an opioid overdose, such as:
- Patients with a history of opioid use disorder
- Those who are currently taking benzodiazepines or other types of central nervous system depressants
- Patients who have already had an opioid overdose
- Those who are living with family members who are at risk for an opioid overdose
- Patients who have children who may be at risk for accidentally swallowing the opioid medication
Opioid Crisis a Priority for Food and Drug Administration
FDA Commissioner Stephen M. Hahn, MD, stated recently that even during the pandemic, the opioid crisis has continued to be a priority. He went on to say that this new recommendation is a step toward raising awareness about naloxone as a “potentially lifesaving treatment” for those at higher risk for opioid overdose. Dr. Hahn explained that all of the tools the FDA has at its disposal will be used to deal with the opioid crisis. He said that efforts to increase access to naloxone have the potential to make an important medication to treat opioid overdose available to those who need it most.
Sources:
empr.com/drug/naloxone-hcl/
fda.gov/drugs/drug-safety-and-availability/fda-recommends-health-care-professionals-discuss-naloxone-all-patients-when-prescribing-opioid-pain
psychiatryadvisor.com/home/topics/addiction/naloxone-recommendations-prescribing-information-label-opioid-use-disorder/